|
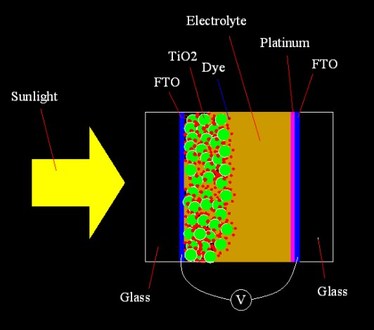
Fig.1 shows the structure of a DSSC.
The dye molecules are adsorbed on the surface of TiO2 nano-particles. When dye molecules absorb the photons of sunlight, the electrons inside the dye molecules are excited and injected into the conductive band of TiO2 nano particles. As all TiO2 particles are connected together forming a porous structure which adhere to the FTO film (Fluorine doped Tin Oxide, a sort of transparent conductive film), the injected electrons will defuse through the TiO2 porous structure to the FTO film. The electrons collected by the FTO film flow to the opposite electrode through the outer circuit, which is driven by the electric potential difference between the two electrodes. On the other hand, after losing an electron to TiO2, the positively charged dye molecule will trap an electron from an iodide ion in the electrolyte to return to its original neutral status, and oxidize the iodide ion into triiodide ion at the same time. The triiodide ion diffuses to the opposite substrate, whereby, catalyzed by the platinum, it turns back into iodide ion by obtaining an electron which flows from TIO2 side through the outer circuit. The cycle described here is repeated for the continuous working of DSSC. |